Diffusion Impedance in a Thin Layer Cell

Requires a Wolfram Notebook System
Interact on desktop, mobile and cloud with the free Wolfram Player or other Wolfram Language products.
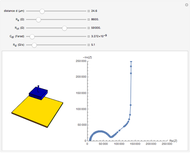
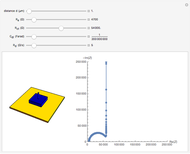
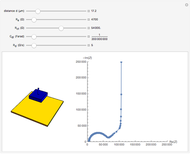
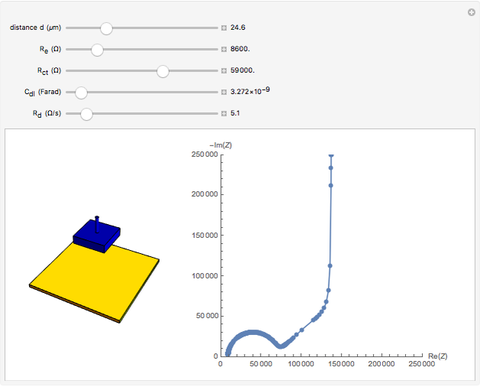
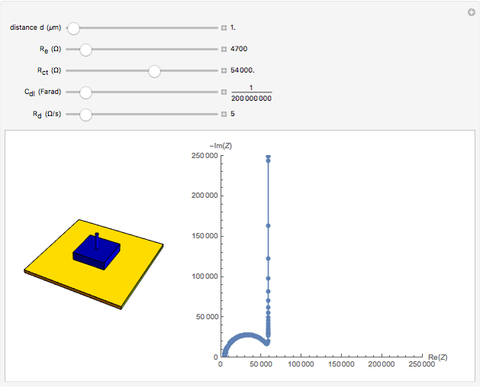
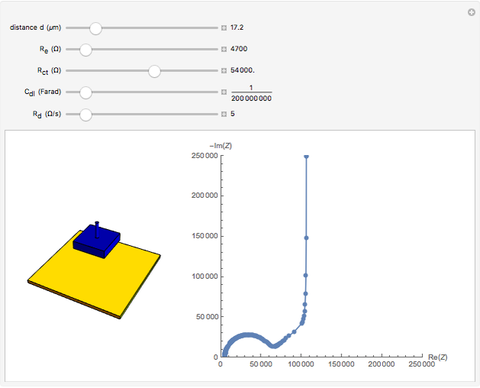
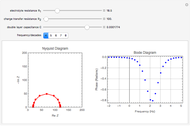
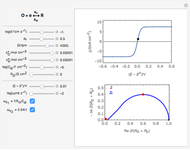
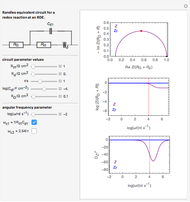
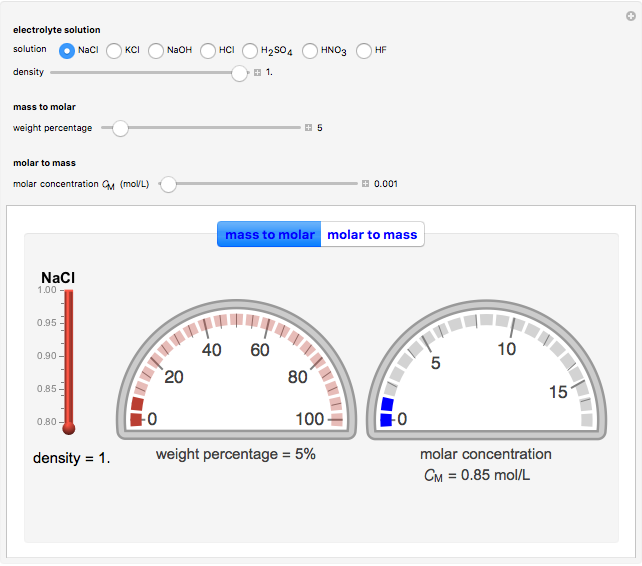
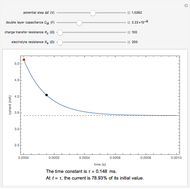
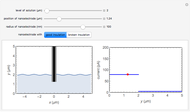
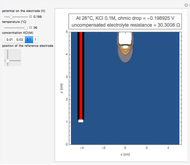
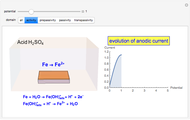
In electrochemistry, a thin layer cell is a device used to perform analytical microanalysis within a thin electrolyte layer. This Demonstration shows the electrochemical diffusion impedance in a thin layer cell. On the left, the configuration of a thin layer cell is shown; the blue plate is the large working electrode, separated from the insulating substrate (green plate) by a distance  (
( m). On the right, the electrochemical impedance is shown in a Nyquist diagram.
m). On the right, the electrochemical impedance is shown in a Nyquist diagram.
Contributed by: Quang-Dao Trinh (June 2011)
Open content licensed under CC BY-NC-SA
Snapshots
Details
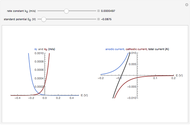
For a thin layer cell of thickness  and the insulating substrate (a boundary impermeable to the diffusing species at the substrate), the diffusion impedance is given [1]:
and the insulating substrate (a boundary impermeable to the diffusing species at the substrate), the diffusion impedance is given [1]:
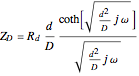 ,
,
where
 is the frequency of the impedance perturbation,
is the frequency of the impedance perturbation,
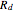 is the scaling factor,
is the scaling factor,
 is the electrolyte film thickness,
is the electrolyte film thickness,
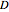 is the diffusion coefficient,
is the diffusion coefficient,
 is the imaginary unit.
is the imaginary unit.
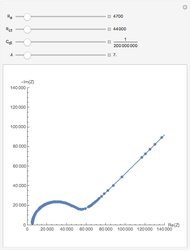
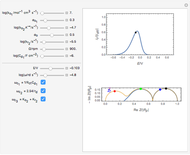
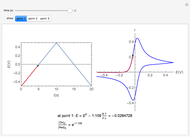
This Demonstration shows that when the electrolyte film thickness  is larger than about 100
is larger than about 100  m, the diffusional impedance behaves like a Warburg impedance (Snapshot 4). For smaller
m, the diffusional impedance behaves like a Warburg impedance (Snapshot 4). For smaller  , however, the impedance at high frequency maintains Warburg behavior, but at low frequencies, it changes to resemble a capacitive impedance (Snapshots 1–3).
, however, the impedance at high frequency maintains Warburg behavior, but at low frequencies, it changes to resemble a capacitive impedance (Snapshots 1–3).
Reference
[1] T. Jacobsen and K. West, "Diffusion Impedance in Planar, Cylindrical and Spherical Symmetry," Electrochimica Acta, 40, 1995 pp. 255–262.
Permanent Citation