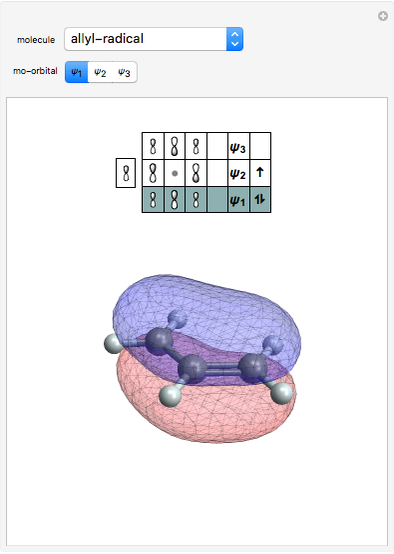
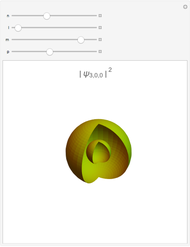
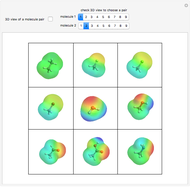
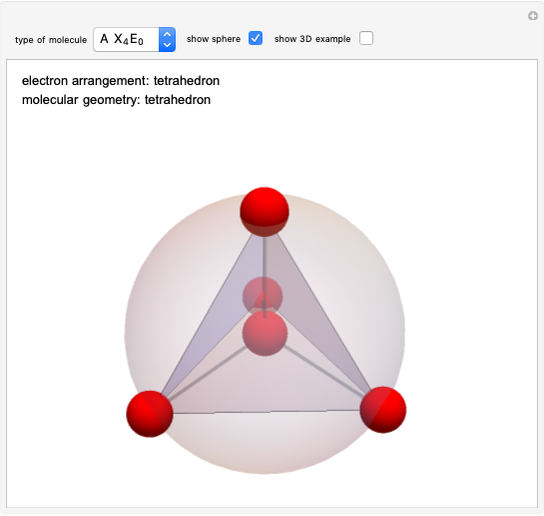
Hybrid Orbitals in Organic Chemistry

Requires a Wolfram Notebook System
Interact on desktop, mobile and cloud with the free Wolfram Player or other Wolfram Language products.
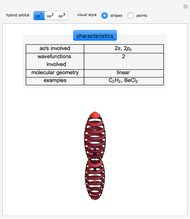
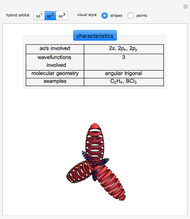
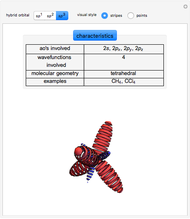
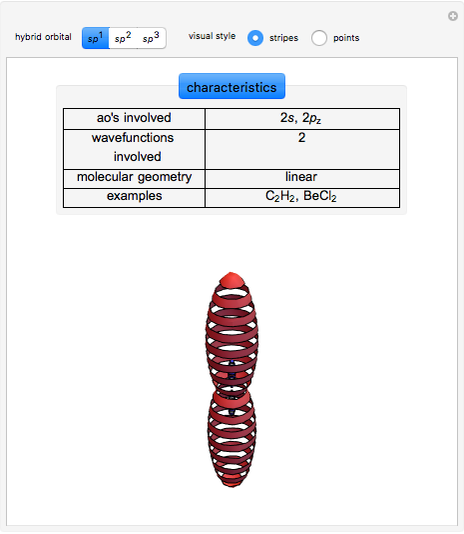
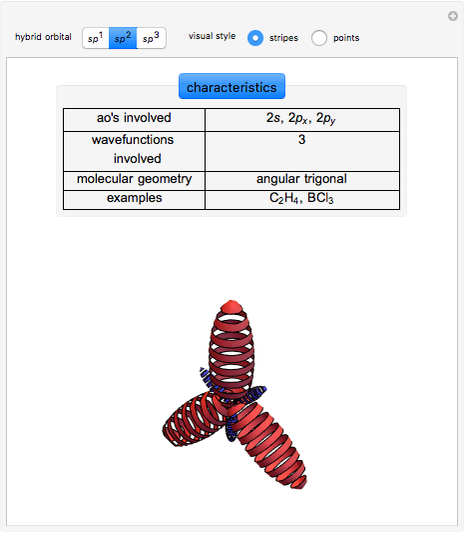
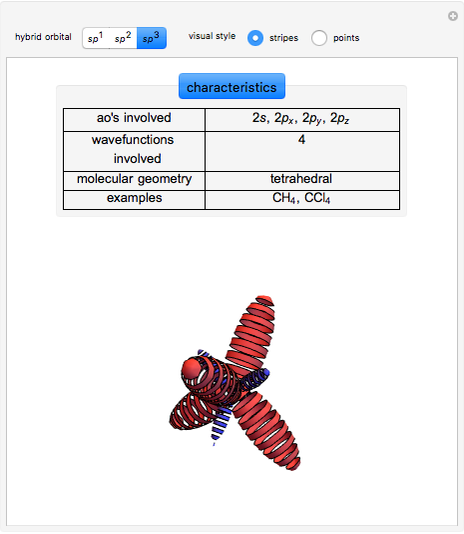
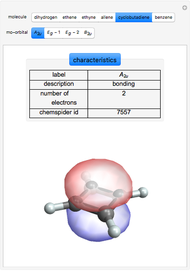
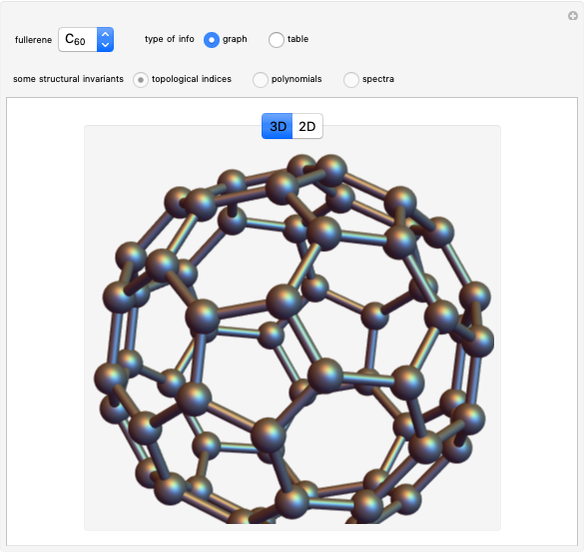
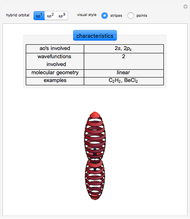
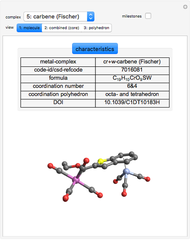
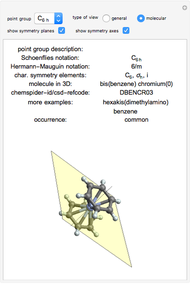
This Demonstration shows the basic characteristics of these hybrid orbitals: the number and type of atomic orbitals involved in the construction, the molecular geometry, examples, and two 3D views (visual style: stripes/points) of the probability density (boundary surface in cartoon form, phase coloring included).
Contributed by: Guenther Gsaller (September 2013)
(Institute of Organic Chemistry, Johannes Kepler University, Linz, Austria)
Open content licensed under CC BY-NC-SA
Snapshots
Details
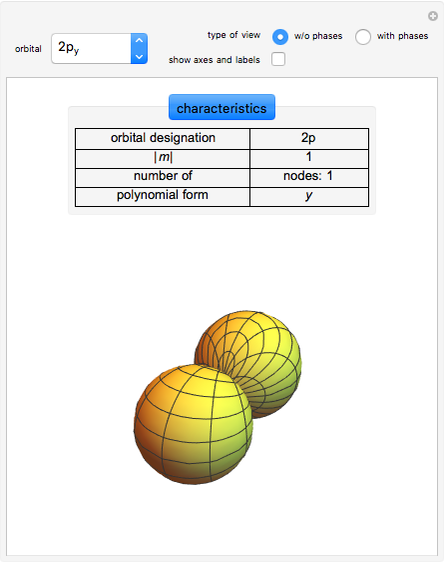
Atomic Orbitals [1]
For given values of  , an atomic orbital can be described by the values
, an atomic orbital can be described by the values 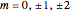 . Angular functions for
. Angular functions for 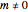 depend on
depend on  ,
,  , or both and are complex. Two complex spherical harmonics
, or both and are complex. Two complex spherical harmonics 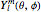 , combined pairwise and linearly, yield real functions. They can be plotted as boundary surfaces.
, combined pairwise and linearly, yield real functions. They can be plotted as boundary surfaces.
The Mathematica function pos inside OrbitalModel is shown at this link: "Problem with SphericalPlot3D plotting". It is used to attach signs to the positive or negative parts of the radial wavefunction, so both parts are colored differently.
This Demonstration shares the code for OrbitalModel and pos with the Demonstration "Visualizing Atomic Orbitals" by the same author.
Hybrid Orbitals
Consider the linear hydride molecule 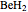 .
.
For Be-H, the following molecule orbitals are formed [2]:
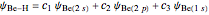 .
.
The 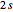 and
and 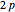 orbitals deliver a new orbital, a so-called hybrid orbital. In the normalized form, the
orbitals deliver a new orbital, a so-called hybrid orbital. In the normalized form, the 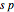 hybrid orbitals can be described by
hybrid orbitals can be described by
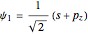 ,
, 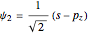 .
.
The normalized hybrid sets (1–3), implemented in this Demonstration, are listed in [3]:
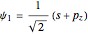 ,
, 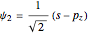 , and
, and 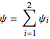
2. 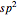 set:
set:
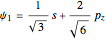 ,
, 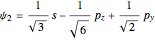 ,
, 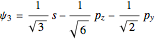 and
and 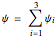
3. 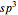 set:
set:
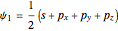 ,
, 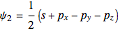 ,
, 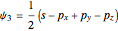 .
.
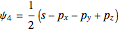 and
and 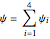 .
.
In [4] a simple method for getting to such sets is presented.
To reduce the size of each graphic produced with SphericalPlot3D, a smaller random sample of its points is taken. ListSurfacePlot3D recreates a striped surface.
The command Graphics3D@Point@Flatten[Cases[Normal[p], Line[x__] :> x was adapted to extract points of various parts of the hybrid orbitals inclusive selective coloring. It is listed at "How can I extract the points from a ListSurfacePlot3D".
References
[1] P. Atkins and R. Friedman, Molecular Quantum Mechanics, Oxford: Oxford University Press, 2011.
[2] D. McQuarrie and J. Simon, Physical Chemistry, Sausalito: University Science Books, 1997.
[3] J. Reinhold, Quantentheorie der Moleküle, Wiesbaden: Springer Fachmedien, 2013. doi: 10.1007/978-3-8348-2037-2.
[4] C. Hsu and M. Orchin, "A Simple Method for Generating Sets of Orthonormal Hybrid Atomic Orbitals," Journal of Chemical Education, 50(2), 1973 pp. 114. doi:10.1021/ed050p114.
Permanent Citation