Pressure Drop in a Packed Bed Reactor (PBR) Using the Ergun Equation

Requires a Wolfram Notebook System
Interact on desktop, mobile and cloud with the free Wolfram Player or other Wolfram Language products.
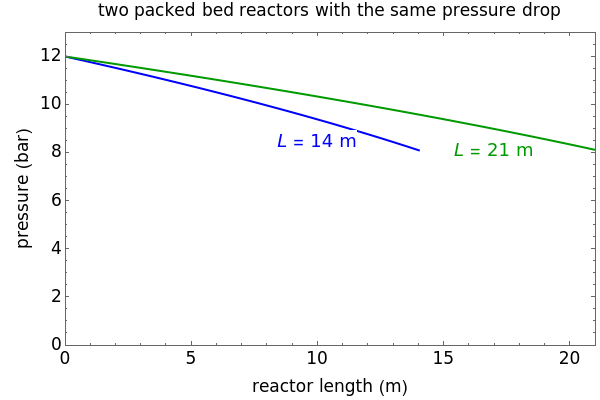
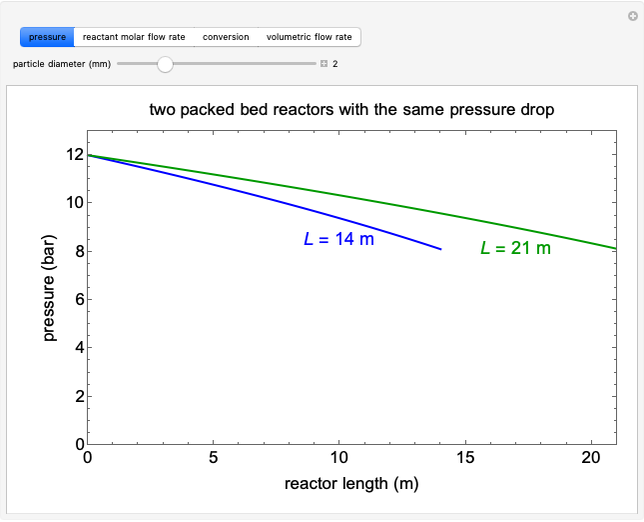
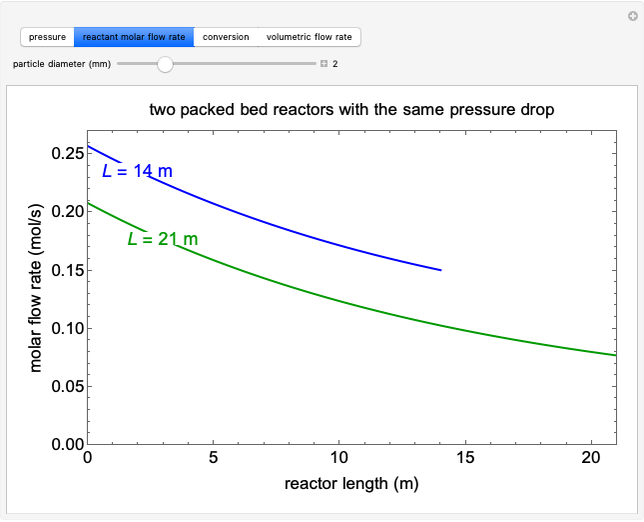
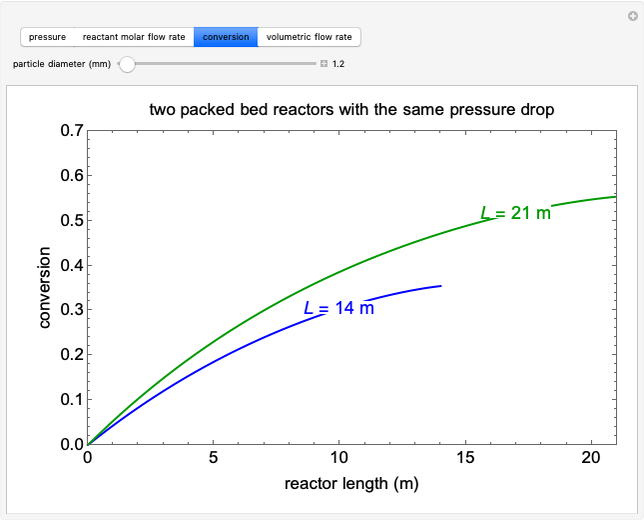
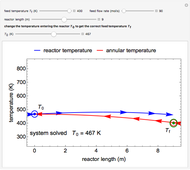
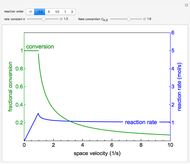
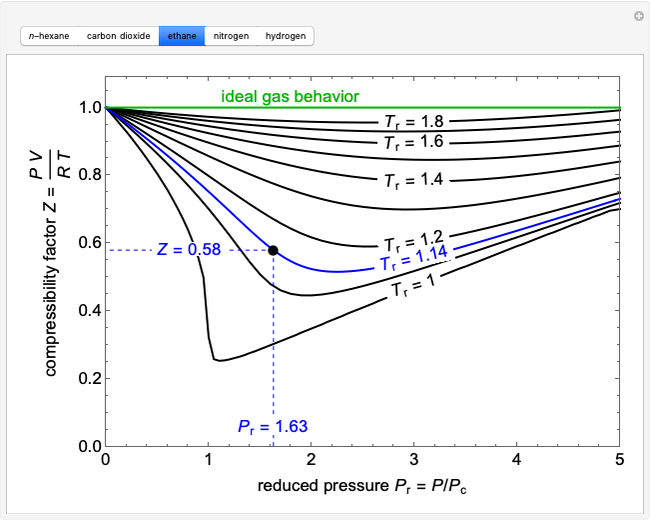
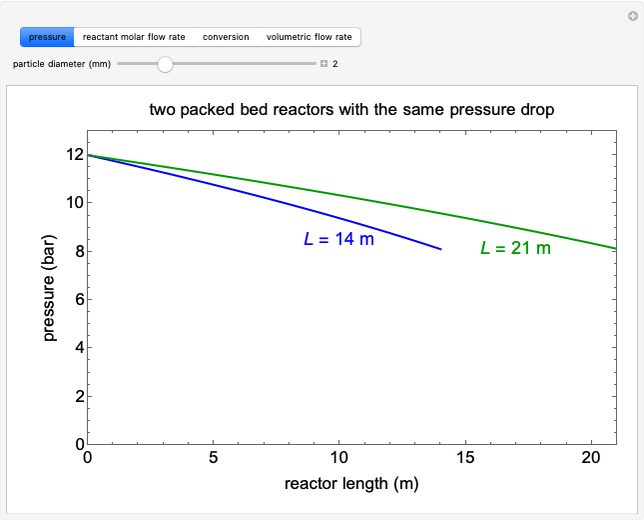
A first-order, irreversible, gas-phase reaction 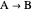 takes place in two isothermal, packed-bed catalytic reactors; they have the same diameter but different lengths (14 and 21 m). Only
takes place in two isothermal, packed-bed catalytic reactors; they have the same diameter but different lengths (14 and 21 m). Only 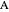 is fed to the reactors. The inlet pressure is the same for each reactor; the outlet pressure changes when particle diameter is changed with the slider, but each reactor has the same outlet pressure. As a result, the inlet molar flow rate of
is fed to the reactors. The inlet pressure is the same for each reactor; the outlet pressure changes when particle diameter is changed with the slider, but each reactor has the same outlet pressure. As a result, the inlet molar flow rate of 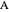 is lower for the longer reactor. Show plots of conversion, pressure, reactant molar flow rate, and volumetric flow rate as a function of distance down the reactor for both reactors with buttons. The Ergun equation is used to model the pressure drop.
is lower for the longer reactor. Show plots of conversion, pressure, reactant molar flow rate, and volumetric flow rate as a function of distance down the reactor for both reactors with buttons. The Ergun equation is used to model the pressure drop.
Contributed by: Rachael L. Baumann (June 2013)
Additional contributions by: John L. Falconer and Nick Bongiardina
(University of Colorado Boulder, Department of Chemical and Biological Engineering)
Open content licensed under CC BY-NC-SA
Snapshots
Details
The pressure decreases down the length of the reactor, and thus the volumetric flow rate increases. As a result, the concentration of the reactant decreases (in addition to the decrease due to conversion), which lowers the rate of reaction.
The Ergun equation for pressure drop in a packed bed reactor (PBR) is:
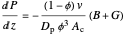 ,
,
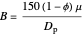 ,
,
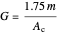 ,
,
where 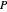 is pressure,
is pressure,  is PBR length,
is PBR length,  is void fraction,
is void fraction,  is volumetric flow rate,
is volumetric flow rate,  is viscosity,
is viscosity, 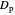 is the diameter of catalyst particles,
is the diameter of catalyst particles, 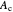 is cross sectional area of the PBR,
is cross sectional area of the PBR, 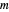 is mass flow rate,
is mass flow rate, 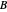 is a laminar flow term, and
is a laminar flow term, and 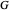 is the turbulent flow term.
is the turbulent flow term.
Material balance on reactant for a first-order reaction:
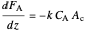 ,
,
where 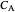 is the molar concentration of reactant
is the molar concentration of reactant 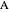 ,
, 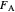 is the molar flow rate of
is the molar flow rate of 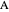 ,
,  is a rate constant, and
is a rate constant, and 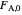 is the inlet molar flow rate of
is the inlet molar flow rate of 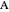 .
.
Since the reaction is 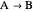 , and only
, and only 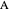 is fed to the reactor, the total molar flow rate
is fed to the reactor, the total molar flow rate 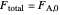 .
.
Volumetric flow rate in reactor (from the ideal gas law):
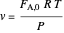 ,
,
where 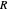 is the ideal gas constant, and
is the ideal gas constant, and 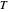 is absolute temperature.
is absolute temperature.
Conversion of reactant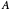 :
:
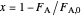 .
.
Permanent Citation