Stellar Nucleosynthesis

Requires a Wolfram Notebook System
Interact on desktop, mobile and cloud with the free Wolfram Player or other Wolfram Language products.
Nearly all matter is composed of "star stuff". According to the Big Bang theory, the early universe was hot enough to allow the nucleosynthesis of hydrogen, helium, and small amounts of lithium and beryllium. Deuterium, a common isotope of hydrogen, was also important as a reactant in many of the reactions required to form helium. The universe expanded and cooled too rapidly to form the heavier elements.
[more]
Contributed by: Jeff Bryant (July 2008)
Open content licensed under CC BY-NC-SA
Snapshots
Details
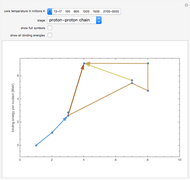
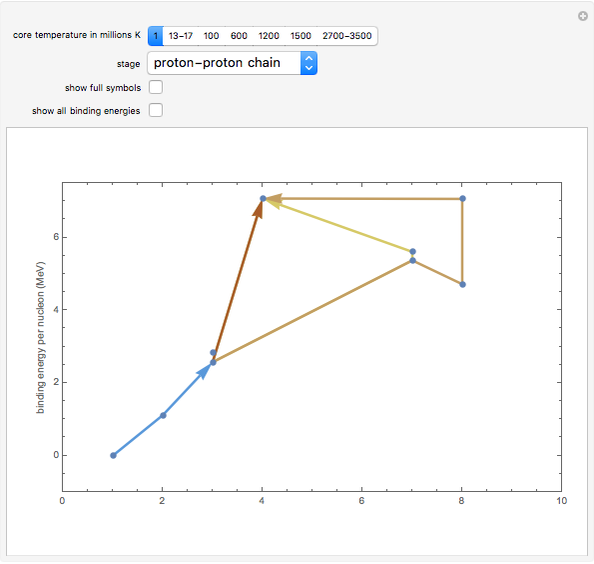
Snapshot 1: stars like our Sun obtain most of their energy from the proton-proton chain, fusing hydrogen in helium
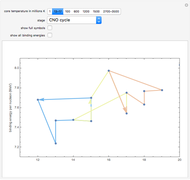
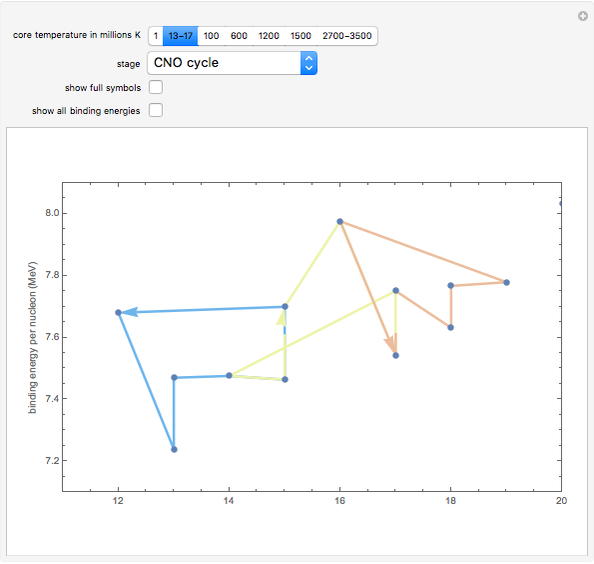
Snapshot 2: stars with slightly hotter cores continue fusing hydrogen, but through reactions that involve carbon, nitrogen, and oxygen (the CNO cycle) created in earlier generations of stars
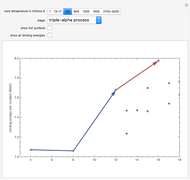
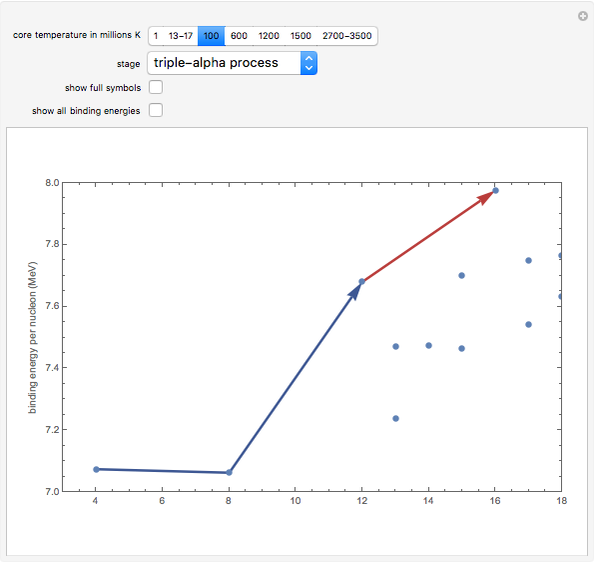
Snapshot 3: Once the hydrogen in the core is used up, the core collapses until it heats enough to ignite the helium ash from earlier reactions. The primary reactions involve three helium nuclei (alpha particles) and result in the formation of carbon and oxygen in the core. Stars begin to swell into red giants at this phase, due to the increased radiation pressure pushing outward from the core.
Snapshot 4: additional alpha reactions, starting with carbon, can also take place and can create neon and magnesium
Snapshot 5: once the helium is used up, the core collapses and heats up again until the carbon ignites; this results in the formation of oxygen, magnesium, sodium, and neon
Snapshot 6: after carbon fusion and additional core collapse, the neon is the next fuel to ignite and results in more oxygen and magnesium formation
Snapshot 7: oxygen fusion processes result in the formation of yet heavier elements such as phosphorus, sulfur, and silicon
Snapshot 8: Finally, the silicon fusion processes come at the end of a very massive star's lifetime and last for only a couple of weeks. This chain rapidly proceeds through the formation of elements such as sulfur, argon, calcium, titanium, chromium, iron, and nickel.
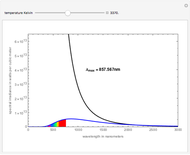
Snapshot 9: Once nickel and iron (from the decay of nickel) are formed in the core, there are no further fusion reactions possible. These elements have the highest binding energy per nucleon of any other elements that are formed, and lie at the peak of this plot. Additional fusion would absorb energy, and so energy generation in the core essentially halts. The core collapses and results in a supernova explosion.
Although stars cannot form elements heavier than iron by standard core fusion, neutron capture during supernova explosions as well as other processes can lead to the heaviest naturally formed elements. These are known as the s- and r-processes, referring to slow and rapid neutron capture. The s-process occurs primarily in old red giants, while the flood of neutrons from a supernova event results in rapid neutron capture events in the r-process.
Permanent Citation
"Stellar Nucleosynthesis"
http://demonstrations.wolfram.com/StellarNucleosynthesis/
Wolfram Demonstrations Project
Published: July 17 2008