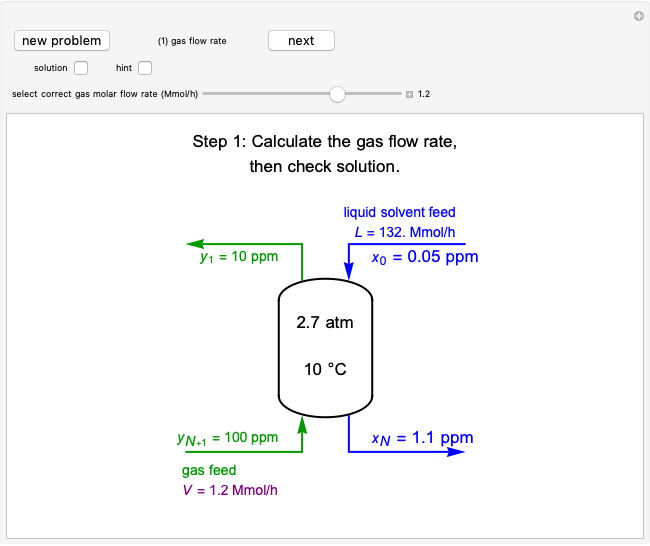
Vapor-Liquid Equilibrium Diagram for Non-Ideal Mixture

Requires a Wolfram Notebook System
Interact on desktop, mobile and cloud with the free Wolfram Player or other Wolfram Language products.
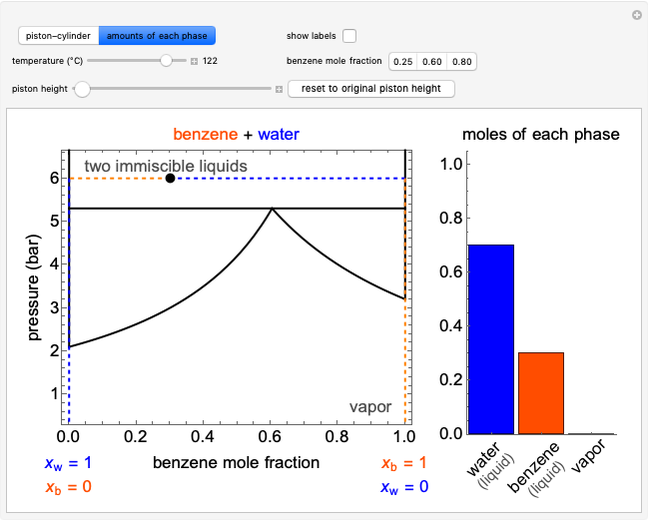
This Demonstration shows 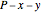 and
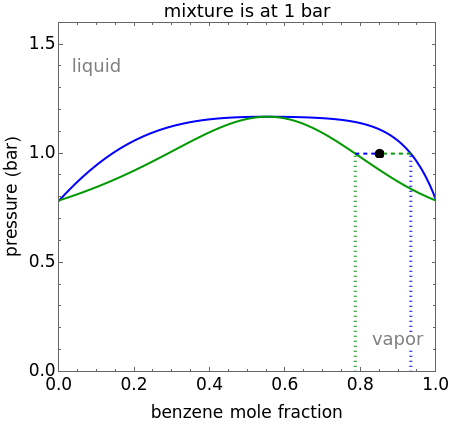
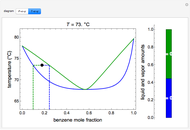
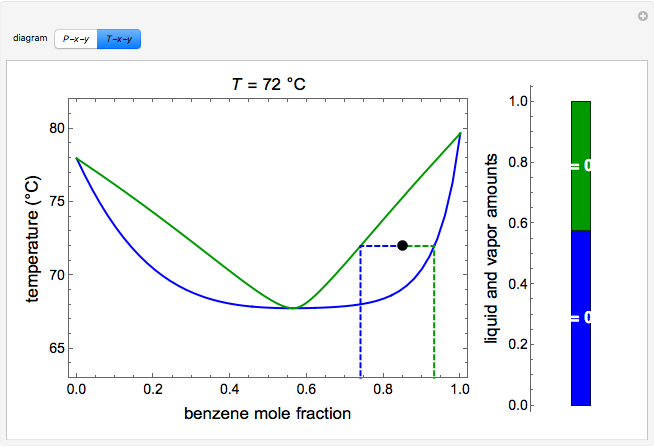
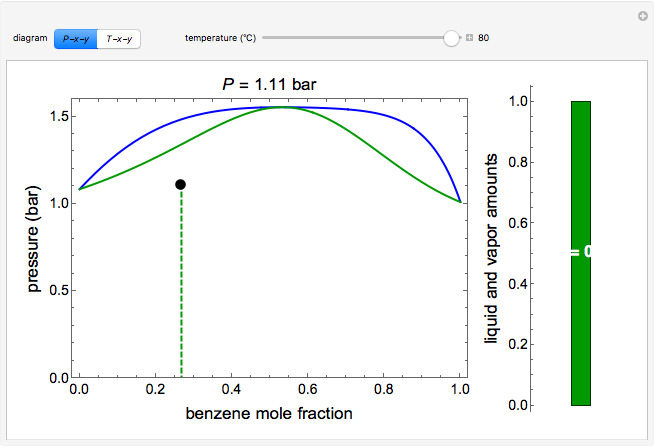
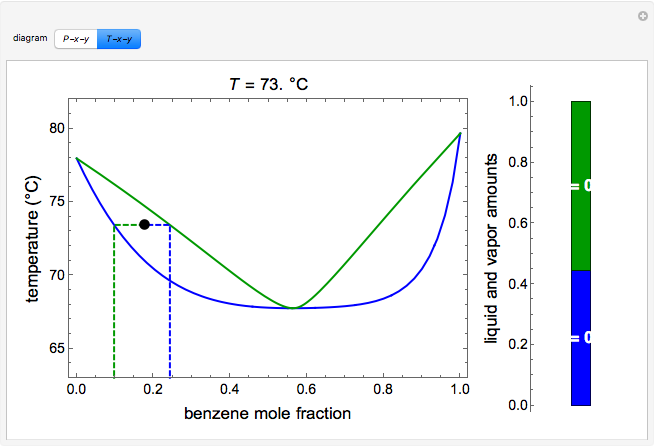
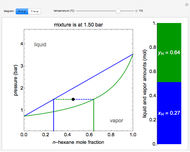
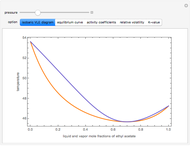
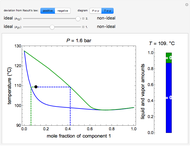
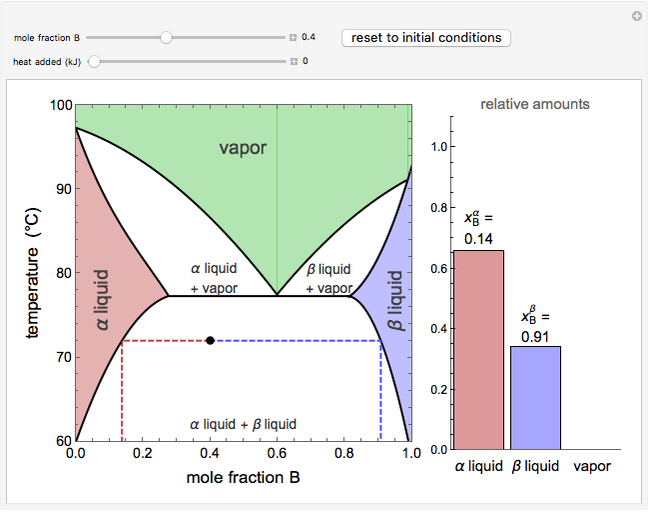
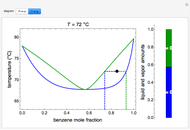
and 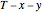 diagrams for vapor-liquid equilibrium (VLE) of a benzene/ethanol mixture. This liquid mixture is non-ideal, and the system has an azeotrope (a condition where liquid and vapor have the same composition). The blue curve represents the liquid-phase boundary (bubble point), and the green curve represents the vapor-phase boundary (dew point). You can vary the temperature for the
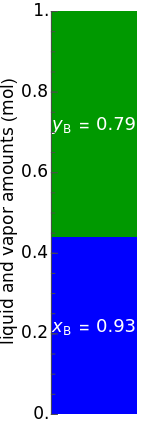
diagrams for vapor-liquid equilibrium (VLE) of a benzene/ethanol mixture. This liquid mixture is non-ideal, and the system has an azeotrope (a condition where liquid and vapor have the same composition). The blue curve represents the liquid-phase boundary (bubble point), and the green curve represents the vapor-phase boundary (dew point). You can vary the temperature for the 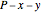 diagram. Click and drag the black dot to change the benzene mole fraction and the temperature or pressure. The bar chart shows the moles of liquid (blue) and vapor (green) in equilibrium and the mole fraction of benzene in each phase (
diagram. Click and drag the black dot to change the benzene mole fraction and the temperature or pressure. The bar chart shows the moles of liquid (blue) and vapor (green) in equilibrium and the mole fraction of benzene in each phase (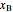 for liquid phase,
for liquid phase, 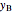 for vapor phase). The system contains a total of 1 mol. The activity coefficients used in the modified Raoult's law are calculated using the two-parameter Margules model.
for vapor phase). The system contains a total of 1 mol. The activity coefficients used in the modified Raoult's law are calculated using the two-parameter Margules model.
Contributed by: Rachael L. Baumann (June 2014)
Additional contributions by: John L. Falconer and Nick Bongiardina
(University of Colorado Boulder, Department of Chemical and Biological Engineering)
Open content licensed under CC BY-NC-SA
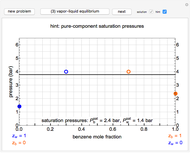
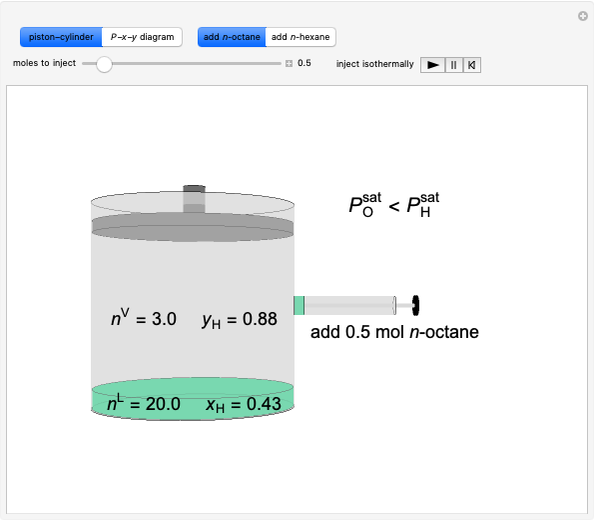
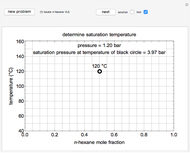
Snapshots
Details
The saturation pressure of component 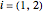 is calculated using the Antoine equation:
is calculated using the Antoine equation:
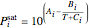 ,
,
where 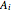 ,
, 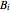 , and
, and 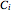 are Antoine constants,
are Antoine constants, 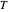 is temperature (°C), and
is temperature (°C), and 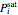 is in bar.
is in bar.
The two-parameter Margules model is used to calculate the activity coefficients for a non-ideal liquid mixture of benzene 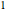 and ethanol
and ethanol  . This model fits the excess Gibbs free energy:
. This model fits the excess Gibbs free energy:
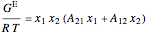 ,
,
where 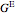 is excess Gibbs energy, and
is excess Gibbs energy, and 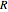 is the ideal gas constant.
is the ideal gas constant.
The activity coefficients 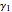 ,
, 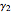 are given by:
are given by:
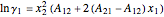 ,
,
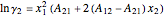 ,
,
where 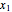 and
and 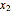 are the liquid mole fractions of benzene and ethanol and
are the liquid mole fractions of benzene and ethanol and 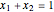 , and
, and 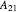 and
and 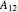 are the Margules parameters.
are the Margules parameters.
The modified Raoult's law is used to calculate the bubble-point and dew-point pressures using the 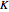 factors:
factors:
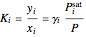 ,
,
where 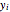 is the vapor mole fraction and
is the vapor mole fraction and 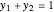 , and
, and  is the total pressure (bar).
is the total pressure (bar).
Bubble-point pressure calculation:
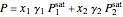 .
.
Dew-point pressure calculation:
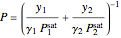 .
.
The screencast video at [2] shows how to use this Demonstration.
References
[1] J. R. Elliott and C. T. Lira, Introductory Chemical Engineering Thermodynamics, New York: Prentice Hall, 2012 pp. 372–377, 430.
[2] Vapor-Liquid Equilibrium Diagram for Non-Ideal Mixture. www.colorado.edu/learncheme/thermodynamics/VLEDiagramNonIdealMixture.html.
Permanent Citation