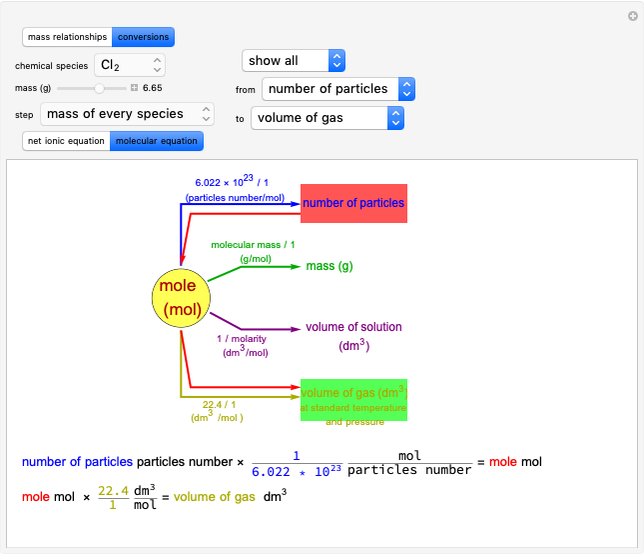
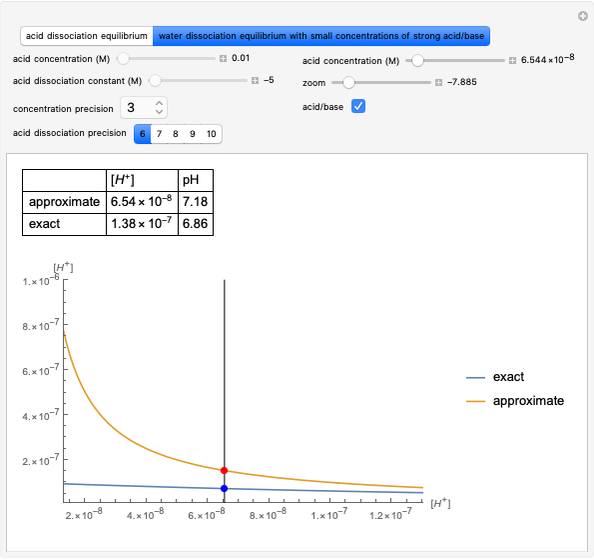
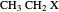Transition States and Intermediates in E1-E1cB-E2 Elimination Reaction Sequence

Requires a Wolfram Notebook System
Interact on desktop, mobile and cloud with the free Wolfram Player or other Wolfram Language products.
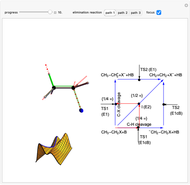
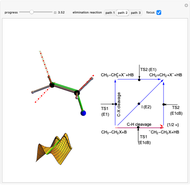
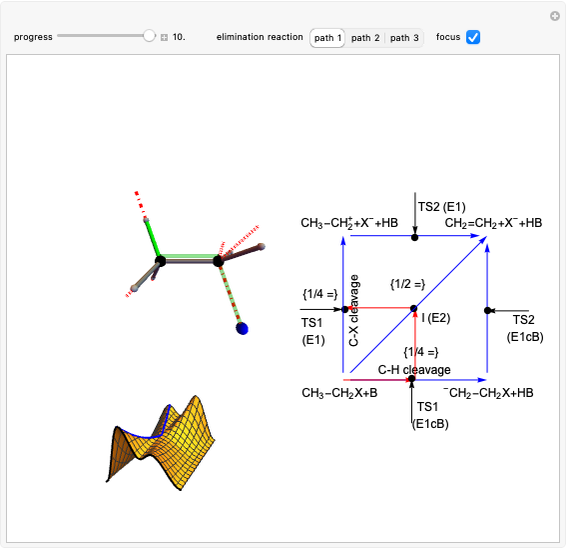
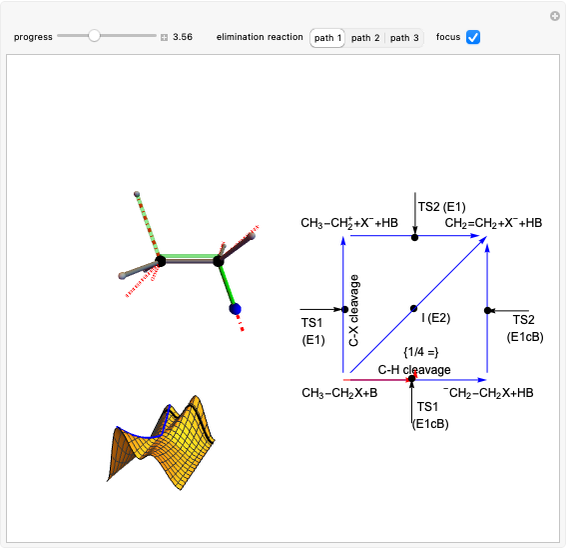
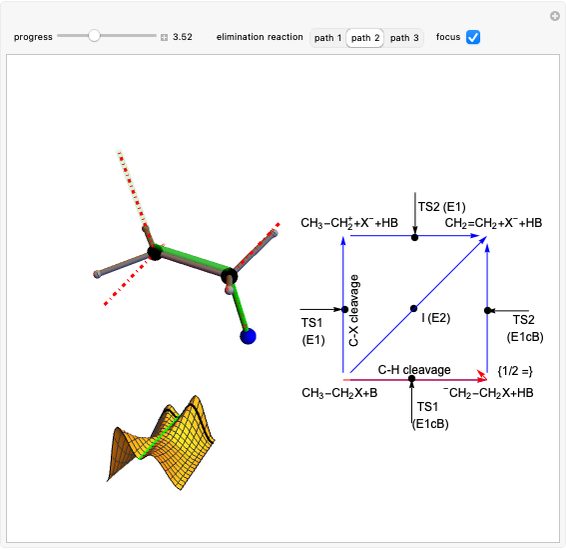
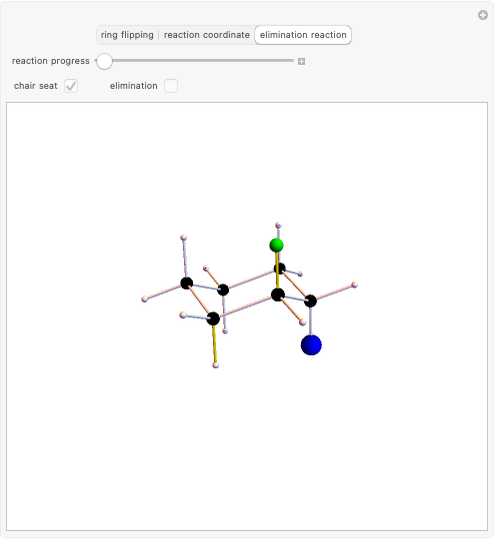
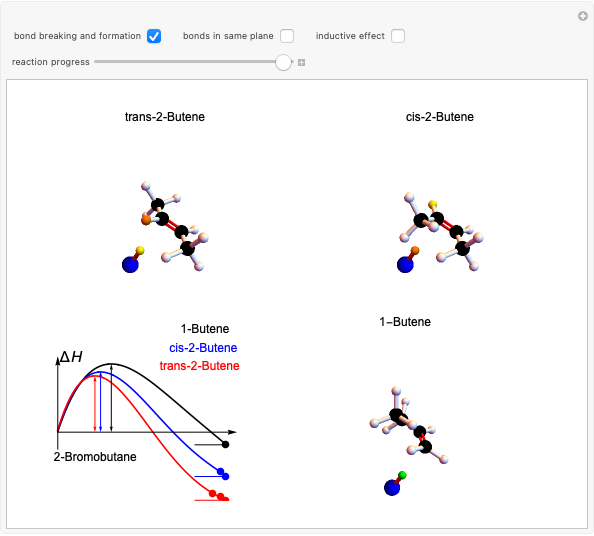
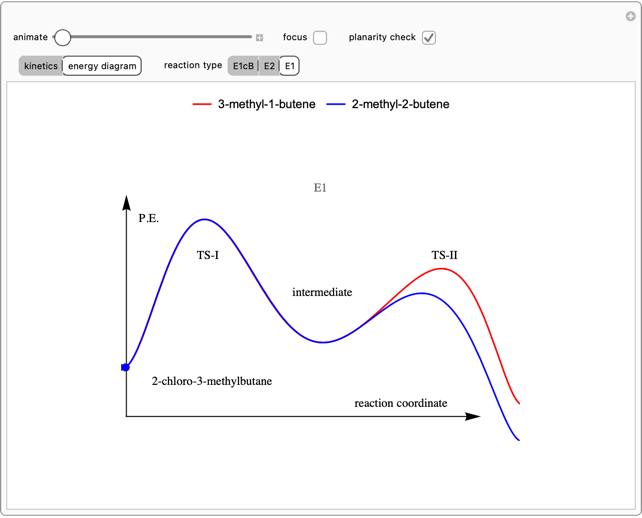
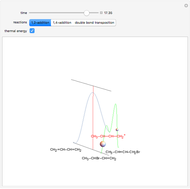
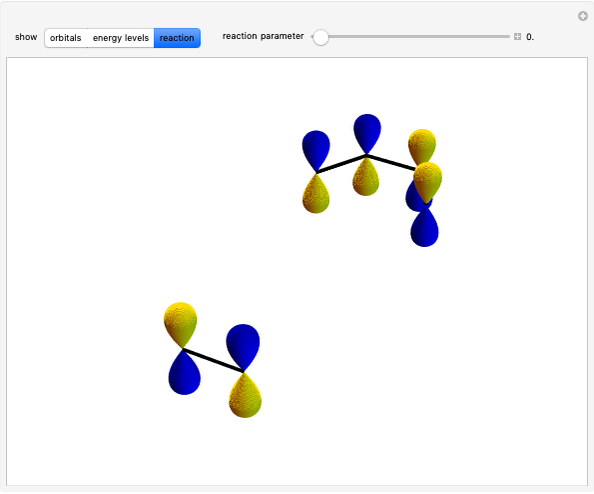
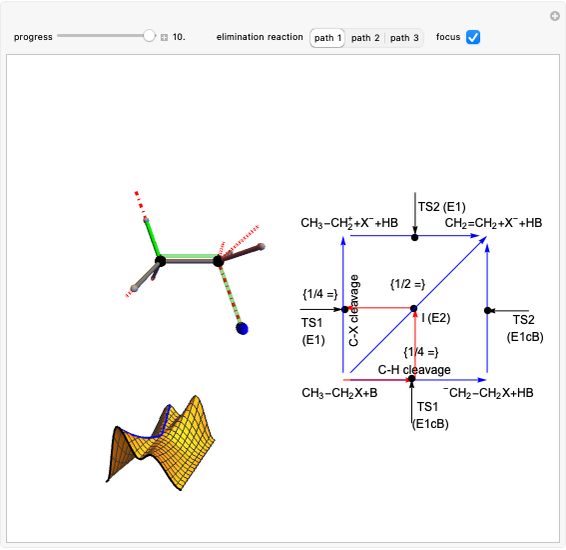
This Demonstration shows various types of elimination reactions for alkyl halides. Three cases can occur, depending on whether the hydrogen in the 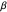 -position detaches earlier (E1cB), simultaneously (E2) or later than the halogen of the molecule (E1). These three possibilities are influenced by the leaving group (halogen). E1cB and E1 reactions take place in two steps, and therefore exhibit two transition states (TS1 and TS2). The E2 reaction takes place in a single step, with a reaction intermediate (I) [1–3].
-position detaches earlier (E1cB), simultaneously (E2) or later than the halogen of the molecule (E1). These three possibilities are influenced by the leaving group (halogen). E1cB and E1 reactions take place in two steps, and therefore exhibit two transition states (TS1 and TS2). The E2 reaction takes place in a single step, with a reaction intermediate (I) [1–3].
Contributed by: D. Meliga, V. Giambrone, L. Lavagnino and S. Z. Lavagnino (August 27)
Open content licensed under CC BY-NC-SA
Details
Snapshot 1: First transition state (TS1) of the E1cB elimination; as the CH bond weakens, the double bond starts forming. The energy in the three-dimensional plot is a minimum at the intersection between the curves.
Snapshot 2: Intermediate state (I) of the E1cB elimination; as the CH bond weakens, the double bond begins to form. The energy is again a minimum at the intersection between the curves.
Snapshot 3: Second transition state (TS2) of the E1cB elimination; as the CH bond weakens, the double bond is almost formed. The energy in the three-dimensional plot is a maximum at the intersection.
References
[1] H. Hart, L. E. Craine and D. J. Hart, Organic Chemistry: A Short Course, 10th ed., Boston: Houghton Mifflin, Co., 1999.
[2] S. Z. Lavagnino, Eliminazione secondo Hofmann [Video]. (Jun 26, 2023) www.youtube.com/watch?v=nZDIQsNCpjM&list=PLswwssc6Q2yYoP_INHmbmouyxW8oP _Gib&index=47.
[3] J. Ashenhurst. "E1cB–Elimination (Unimolecular) Conjugate Base." Master Organic Chemistry. (Jun 26, 2023) www.masterorganicchemistry.com/2020/02/11/e1cb-elimination-unimolecular-conjugate-base.
Snapshots
Permanent Citation